About Sernova, Corp. À propos de Sernova, Corp.
Sernova is committed to the development and clinical advancement of its products for metabolic, hematological and other chronic diseases using therapeutic cells transplanted into a patented implanted medical device, which forms an organ-like environment promoting long-term function and survival of the therapeutic cells. Sernova s'engage dans le développement et l'avancement clinique de ses produits contre les maladies métaboliques, hématologiques et autres maladies chroniques utilisant des cellules thérapeutiques transplantées dans un dispositif médical implanté breveté, qui forme un environnement semblable à un organe favorisant la fonction et la survie à long terme des cellules thérapeutiques.
The company’s management believes in building strong and long-lasting collaborations and partnerships that would lead to the rapid advancement of Sernova’s portfolio of products into the market, improving global health and bringing value to patients and society, in concert with our clinical development programs. La direction de la société croit à la mise en place de collaborations et de partenariats solides et durables qui permettraient au portefeuille de produits de Sernova de progresser rapidement sur le marché, d’améliorer la santé mondiale et d’apporter une valeur ajoutée aux patients et à la société, de concert avec nos programmes de développement clinique.
Indications: Les indications:
Diabetes Diabète
Haemophilia A Hémophilie A
Thyroid disease Maladie thyroïdienne
Clinical studies: Etudes cliniques:
Diabetes US phase I/II clinical study cleared by the FDA Etude clinique de phase I / II sur le diabète, approuvée par la FDA
First-in-human study in Diabetic subjects with hypoglycemia unawareness Première étude chez l'homme chez des sujets diabétiques peu conscients de l'hypoglycémie
Cell Pouch System™

Sernova’s Cell Pouch System™ is a novel implantable and scalable medical device which forms a highly vascularized organ-like environment in the body for the housing, function and long-term survival of therapeutic cells. These therapeutic cells release necessary proteins or hormones missing from the body to treat chronic diseases as an alternative to daily administration of drugs. Le Cell Pouch System™ est un nouveau dispositif médical préalablement implanté et formant un environnement naturel hautement vascularisé afin d’y loger des cellules thérapeutiques, favorisant leur bon fonctionnement et leur survie dans le corps. Ces cellules thérapeutiques libèrent les protéines ou les hormones nécessaires pour traiter les maladies chroniques comme alternative a l’administration quotidienne de médicaments.

Immune Protection Protection immunitaire
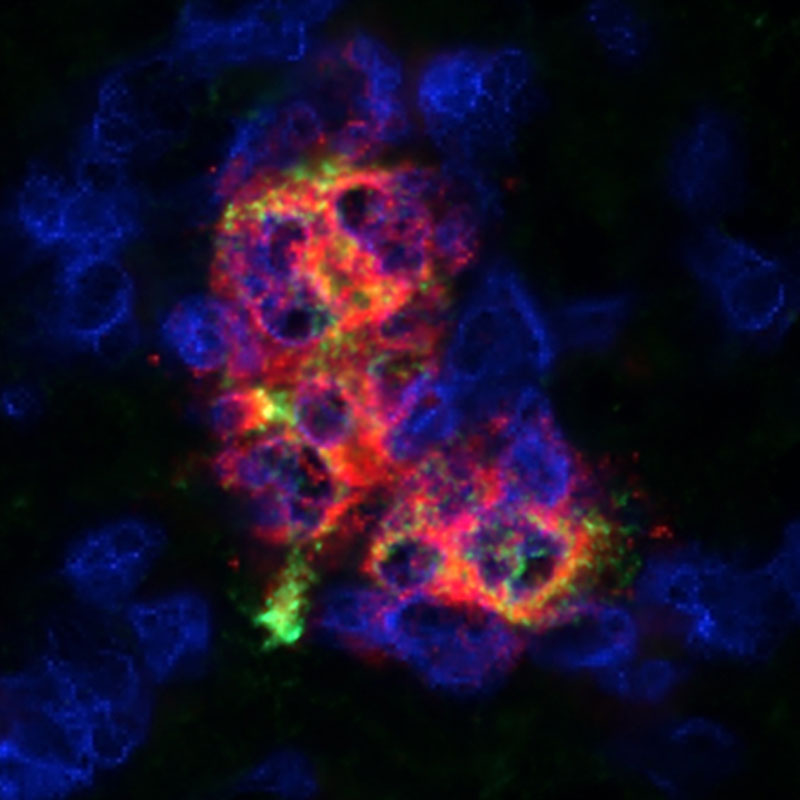
We have shown that cells can be protected using medications that prevent immune system attack within the Cell Pouch™. Nous avons montré que les cellules peuvent être protégées en utilisant des médicaments qui empêchent les attaques du système immunitaire dans la Cell Pouch™.
Microencapsulation technologies house cells within the Cell Pouch chambers and protect the cells from immune system attack. Les technologies de microencapsulation hébergent les cellules dans les chambres à cellules et les protègent des attaques du système immunitaire.
Technologies are in development to make transplanted cells unrecognizable to the immune system. Des technologies sont en cours de développement pour rendre les cellules greffées méconnaissables du système immunitaire.
Sernova’s Cell Pouch™, combined with immune protected therapeutic cells, offers protection from immune system attack creating an effective, safe, long-term and convenient therapeutic option for patients with chronic diseases who seek to improve their quality of life. Le Cell Pouch™ de Sernova, associé à des cellules immunitaires protégées, offre une protection contre le système immunitaire avec une option thérapeutique efficace, sûre, à long terme et pratique pour les patients atteints de maladies chroniques qui cherchent à améliorer leur qualité de vie.

Featured News Nouvelles en vedette
Sernova Business Update
Marek Sutherland of CTV News London - Cure for type one diabetes getting closer, London company says
Noah Stansfield of CGT Live - Patients With T1D Achieve Insulin Independence Following Implantation of Cell Pouch System and Islet Transplant
Sean Whooley and Danielle Kirsh of Fast Five- Teleflex has a Class I recall, Boston Scientific appoints two new board directors (Sernova discussed in the podcast recording from 1:00-2:58 time marks)
Lei Lei Wu of EndPoints News - Sernova says five diabetes patients have now been taken off insulin after 'cell pouch' therapy
Shane Whooley of MassDevice - Sernova reports positive interim data for Cell Pouch System
News Releases Communiqués de presse
Sernova Announces AGM Voting Results
Sernova Provides Organizational Update
Sernova Welcomes Dr. Bernd Muehlenweg as Evotec’s Nominee for its Board of Directors
Sernova Provides Positive Clinical and Platform Portfolio Update
Updates Mises à jour
Sernova KOL Event on Thyroid Disease with Dr. Sam Wiseman
Watch now!
Visionner maintenant!
Events Événements
Sernova AGM - Annual General Meeting Sernova AGM - Annual General Meeting
2024 Bloom Burton & Co. Healthcare Investor Conference 2024 Bloom Burton & Co. Healthcare Investor Conference
Oppenheimer 34th Annual Healthcare Medtech & Services Conference Sernova Presentation at 1:20-1:50pm EST. Oppenheimer 34e conférence annuelle sur les technologies médicales et les services de santé Présentation Sernova à 13h20-13h50 HNE.
Invest with Sernova Investir dans Sernova
If you are a shareholder, investor, broker, analyst, journalist, investment advisor, or looking to develop business opportunities, please feel free to contact us by email or telephone. Si vous êtes actionnaire, investisseur, courtier, analyste, journaliste, conseiller en placement ou souhaitez développer des opportunités d’affaires, n'hésitez pas à nous contacter par email ou par téléphone.
Sernova is a regenerative medicine company developing therapeutic technologies with multibillion-dollar market potential for each of its clinical indications. Sernova est une société de médecine régénérative développant des technologies thérapeutiques offrant un potentiel de marché de plusieurs milliards de dollars pour chacune de ses indications cliniques.
Sernova is a Collaborative Team Sernova, c’est aussi les collaborations
We believe in advancing our clinical programs and building strong and long-lasting collaborations and partnerships that will lead to the rapid advancement of Sernova’s portfolio of products into the market, improving global health and bringing value to patients and society. Nous croyons en la promotion de nos programmes cliniques et en la mise en place de collaborations et de partenariats solides et durables qui permettront au portefeuille de produits de Sernova de progresser rapidement sur le marché, d'améliorer la santé mondiale et d'apporter de la valeur aux patients et à la société.
Privacy PolicyPolitique de confidentialité
Updated July 6, 2018
Please read this Policy carefully along with our Legal Notice that describes our Terms of Use for the Website.
By accessing www.sernova.com (the “Website”) you hereby agree with the practices described in this Privacy Policy (the “Policy”)
This Policy applies to all information gathered through the Website and/or any related marketing technique or events.
Information Collection
The information collected is limited to the information that you decide to share with us through the News Dispatch Service, when participating at event or activities or in the general course of business by expressing an interest in obtaining information about Sernova Corp. and our products, such as name, email, phone number, and similar contact information. This information is stored through MailChimp (please refer to MailChimp Privacy Policy at https://mailchimp.com/legal/privacy/).
Information Sharing
Sernova Corp. is the sole owner of any information collected on the Website. We do not sell, share or rent this information to others.
Traffic and Automatic Information Collection
Sernova Corp. maintains log files of the traffic on www.sernova.com. This information is not linked to any personal information that you have provided us. Logs are used to manage traffic, identify content accessed, and IT requirements. Information logged and automatically collected includes without being limited to IP addresses and browser types. This information does not reveal your specific identity.
Cookies
Cookies can be used to provide you with a more personalized experience. The Website may use cookies to make that experience more companionable when you return to the Website. You have the option at all time to decline the use of cookies. If you choose to do so, you may not be able to fully use all features of the Website. You can also delete cookie files at all time from your computer. Those cookies may include first-party cookies (such as the Google Analytics cookies).
Updates
This Policy is a living document and may be amended or updated from time to time without further notice. We encourage you to review the Policy periodically.
Contact
If you have any questions or comments about our policy, you can email us at info@sernova.com or by phone at 1(877) 299-4603 or by mail at
Sernova Corp.
700 Collip Circle, Suite 114
London, ON Canada N6G 4X8
Mis à jour le 6 juillet 2018
Veuillez lire attentivement cette politique ainsi que notre avis juridique qui décrit nos conditions d'utilisation du site Web.
En accédant à www.sernova.com (le «site Web»), vous acceptez les pratiques décrites dans la présente politique de confidentialité (la «politique»).
Cette politique s'applique à toutes les informations collectées via le site Web et / ou toute technique ou événement marketing associé.
Collecte d'informations
Les informations collectées se limitent aux informations que vous décidez de partager avec nous par le biais du service d’expédition de nouvelles, lorsque vous participez à un événement ou à des activités ou que vous vous intéressez à obtenir des informations sur Sernova Corp. comme nom, email, numéro de téléphone et informations de contact similaires. Ces informations sont stockées via MailChimp (veuillez vous reporter aux règles de confidentialité de MailChimp sur https://mailchimp.com/legal/privacy/).
Partage d'information
Sernova Corp. est l'unique propriétaire de toute information collectée sur le site Web. Nous ne vendons pas, ne partageons pas ou ne louons pas ces informations à des tiers.
Collecte d'informations routières et automatiques
Sernova Corp. gère les fichiers journaux du trafic sur www.sernova.com. Ces informations ne sont liées à aucune information personnelle que vous nous avez fournie. Les journaux sont utilisés pour gérer le trafic, identifier le contenu accédé et les besoins informatiques. Les informations consignées et collectées automatiquement ne sont pas limitées aux adresses IP et aux types de navigateur. Cette information ne révèle pas votre identité spécifique.
Cookies
Les cookies peuvent être utilisés pour vous offrir une expérience plus personnalisée. Le site Web peut utiliser des cookies pour rendre cette expérience plus conviviale lorsque vous revenez sur le site Web. Vous avez la possibilité à tout moment de refuser l'utilisation de cookies. Si vous choisissez de le faire, vous ne pourrez peut-être pas utiliser toutes les fonctionnalités du site Web. Vous pouvez également supprimer des fichiers de cookies à tout moment depuis votre ordinateur. Ces cookies peuvent inclure des cookies de première partie (tels que les cookies de Google Analytics).
Mises à jour
Cette politique est un document évolutif et peut être modifié ou mis à jour de temps à autre sans préavis. Nous vous encourageons à consulter la politique périodiquement.
Contact
Si vous avez des questions ou des commentaires sur notre politique, vous pouvez nous envoyer un courriel à info@sernova.com ou par téléphone au 1 (877) 299-4603 ou par courrier à
Sernova Corp.
700 Collip Circle, Suite 114
London, ON Canada N6G 4X8
Press Release Communiqué de presse - Mai 01, 2024 1 Mai, 2024
Sernova Announces AGM Voting Results
SERNOVA ANNOUNCES AGM VOTING RESULTS
LONDON, Ontario; BOSTON, Massachusetts – May 1, 2024, Sernova Corp. (“Sernova” or the “Corporation”)
(TSX:SVA) (OTCQB:SEOVF) (FSE/XETRA:PSH) a clinical-stage biotechnology company focused on the
development of regenerative medicine cell therapies for treatment of chronic diseases, is pleased to announce
results from its Annual General Meeting of Shareholders (the “AGM”) held virtually via live audio webcast, on
April 30, 2024. At the Meeting, a total of 88,316,160 common shares were voted, representing 29.11% of the
votes attached to all outstanding common shares as of the record date. The voting results were as follows:
| Director | % of Votes For | % of Votes Against |
| Cynthia Pussinen | 95.62% | 4.38% |
| James T. Parsons | 98.63% | 1.37% |
| Dr. Steven Sangha | 96.43% | 3.57% |
| Brett A. Whalen | 73.91% | 26.09% |
Accordingly, Cynthia Pussinen, James T. Parsons, Dr. Steven Sangha and Brett A. Whalen were elected to the
board of directors for the ensuing year. In addition, Bernd Muehlenweg was appointed to the board of directors
following the AGM. Bertram von Plettenberg resigned from the board of directors prior to the AGM and did not
stand for election at the AGM.
Shareholders also approved (with 99.79% of the votes approving) the re-appointment of KPMG LLP, Chartered
Professional Accountants as the Company’s auditor for the ensuing year, and approved (with 82.57% of the
votes approving) the amendments to the Option Plan and DSU Plan and the increase in the maximum number of
Common Shares reserved for issuance.
ABOUT SERNOVA AND ITS CELL POUCH SYSTEM PLATFORM FOR CELL THERAPY
Sernova Corp. is a clinical-stage biotechnology company that is developing therapeutic cell technologies for
chronic diseases, including insulin-dependent diabetes, thyroid disease, and blood disorders that include
hemophilia A. Sernova is currently focused on developing a ‘functional cure’ for insulin-dependent diabetes
with its lead technology, the Cell Pouch System, a novel implantable and scalable medical device with immune
protected therapeutic cells.
On implantation, The Cell Pouch forms a natural, vascularized tissue environment in the body allowing long-
term survival and function of therapeutic cells that release essential factors that are absent or deficient in the
bodies of patients with certain chronic diseases. Sernova’s Cell Pouch System has demonstrated its potential to
be a ‘functional cure’ for people with T1D in an ongoing Phase 1/2 clinical study at the University of Chicago.
Sernova partnered with Evotec to develop an implantable o-the-shelf iPSC (induced pluripotent stem cells)
based islet replacement therapy. This partnership provides Sernova a potentially unlimited supply of insulin-
producing cells to treat millions of patients with insulin-dependent diabetes (type 1 and type 2). Sernova’s
development pipeline that uses its Cell Pouch System also includes: a cell therapy for hypothyroid disease
resulting from thyroid gland removal and an ex vivo lentiviral Factor VIII gene therapy for hemophilia A.
FOR FURTHER INFORMATION, PLEASE CONTACT:
Christopher Barnes
VP, Investor Relations
Sernova Corp.
christopher.barnes@sernova.com
519-902-7923
www.sernova.com
FORWARD-LOOKING INFORMATION
This release contains statements that, to the extent they are not recitations of historical facts, may constitute
“forward-looking statements” that involve various risks, uncertainties, and assumptions, including, without
limitation, statements regarding the prospects, plans, and objectives of the company. Wherever possible, but
not always, words such as "expects", "plans", "anticipates", "believes", "intends", "estimates", "projects",
"potential for" and similar expressions, or that events or conditions "will", "would", "may", "could" or "should"
occur are used to identify forward-looking statements. These statements reflect management’s beliefs with
respect to future events and are based on information currently available to management on the date such
statements were made. Many factors could cause Sernova’s actual results, performances or achievements to
not be as anticipated, estimated or intended or to dier materially from those expressed or implied by the
forward-looking statements contained in this news release. Such factors could include, but are not limited to,
the company’s ability to secure additional financing and licensing arrangements on reasonable terms, or at all;
ability to conduct all required preclinical and clinical studies for the company’s Cell Pouch System and or
related technologies, including the timing and results of those trials; ability to obtain all necessary regulatory
approvals, or on a timely basis; ability to in-license additional complementary technologies; ability to execute
its business strategy and successfully compete in the market; and the inherent risks associated with the
development of biotechnology combination products generally. Many of the factors are beyond our control,
including those caused by, related to, or impacted by the novel coronavirus pandemic. Investors should consult
the company’s quarterly and annual filings available on www.sedarplus.ca for additional information on risks
and uncertainties relating to the forward-looking statements. Sernova expressly disclaims any intention or
obligation to update or revise any forward-looking statements, whether as a result of new information, future
events or otherwise.
Press Release Communiqué de presse - Avril 25, 2024 25 Avril, 2024
Sernova Provides Organizational Update
Sernova Provides Organizational Update
• Dr. Philip Toleikis to retire as of April 30, 2024
• Cost savings from restructuring and strategic transformation anticipated to extend runway
• Board rebuild underway
LONDON, Ontario; BOSTON, Massachusetts – April 25, 2024 – Sernova Corp. (TSX:SVA) (OTCQB:SEOVF)
(FSE/XETRA:PSH), a clinical-stage biotechnology company focused on the development of regenerative
medicine cell therapies for treatment of chronic diseases, today announced the retirement of Chief Technology
Officer (CTO), Dr. Philip Toleikis along with progress on strategic transformation efforts including a
restructuring of operations and a workforce reduction of approximately 35%.
Commenting on his retirement, Dr. Toleikis said, “It has been a tremendous honor and privilege to serve
Sernova since 2009, initially as the President, CEO and director, and more recently as its CTO. Significant
progress has been made and there is exciting work ahead that I am confident will be realized by the team,
under the guidance of our CEO, Ms. Pussinen.” Dr. Toleikis further states, “I am pleased to provide continued
support to Sernova as a consultant. I am proud of the Company’s advancements towards redefining the way
chronic diseases will be treated using a cell therapy-based approach while importantly contributing to
improving the lives of those living with chronic diseases.”
“Philip’s contributions to Sernova’s research and development, and fund-raising efforts have been foundational
to the Company’s evolution into a next-generation regenerative medicine company,” said Brett Whalen,
Chairman of the Sernova Board. “He will leave a company which is well-positioned to foster the next wave of
innovations in stem cell therapies. On behalf of the Board of Directors and the entire company, I would like to
thank Dr. Philip Toleikis for his contributions and unwavering dedication to the Company. We wish him all the
best for his future.”
As communicated in early April, following a review of the company’s therapeutic pipeline and emerging
opportunities for its Cell Pouch system platform technologies, Sernova confirmed key priorities including its
lighthouse program in insulin dependent Type 1 Diabetes plus its intention to advance an IND filing for its post-
operative hypothyroidism program. “The pipeline and platform review and associated strategy refresh
highlighted several ways by which to optimize financial resources, extending the Company’s cash runway. We
continue to seek ways to raise additional capital to strengthen our financial foundation,” said Cynthia Pussinen,
Chief Executive Officer of Sernova. “In connection with the strategic transformation, we will pause on any new
investments into the conformal coating program to reallocate funds. In parallel, we continue to evaluate
alternative approaches to obviate the need for immunosuppressive regimens for our allogeneic therapies.”
Sernova has implemented a plan to fortify the balance sheet and cash position, including a workforce
restructuring, representing a key step towards streamlining the organization while ensuring the ability to secure
core competencies needed to drive further progress in key clinical and pre-clinical assets. “Decisions to scale
back or to rebalance headcount are extremely difficult. I want to express my heartfelt appreciation and
gratitude to those impacted for their invaluable contributions, dedication to helping patients and impact to
furthering the Company’s vision of a future where chronic conditions are no longer insurmountable obstacles”,
said Ms. Pussinen.
The Company confirms the completion and closure of all internal investigation efforts, previously announced
with respect to its former CFO and potentially a second employee of the Company. No new findings were
revealed and there will be no further action on the matter. The investigation confirmed that there have been no
securities violations and that findings bore no material impact to financial statements and operations.
Lastly, Sernova Director Mr. Bertram von Plettenberg has retired from the Company’s Board, effective April
23rd. “The Board and Management sincerely thank Mr. von Plettenberg for his past year of service to the
Company, and wish him well in future endeavors,” said Ms. Pussinen. “The evolution and composition of the
Board will continue, as we look to enhance our life sciences industry specific strength, to better serve our
patients and shareholders.”
ABOUT SERNOVA’S ANNUAL GENERAL MEETING
Sernova’s Annual General Meeting will be held on Tuesday April 30, 2024 starting at 1:00 PM ET. The meeting
will be conducted virtually, via live webcast and accessible online at https://virtualmeetings.tsxtrust.com/1571.
Please note that this site may not be fully accessible on all internet browsers, and it is advisable to use a
browser other than Internet Explorer for optimal experience. If you are unable to join the meeting through your
usual web browser, we suggest trying an alternate browser.
For investors who wish to submit questions via the TSX Trust portal voting control numbers will be needed. For
attendees who would simply like to listen to the meeting conduct, you can register as a guest.
ABOUT SERNOVA AND ITS CELL POUCH SYSTEM PLATFORM FOR CELL THERAPY
Sernova Corp. is a clinical-stage biotechnology company that is developing therapeutic cell technologies for
chronic diseases, including insulin-dependent diabetes, thyroid disease, and blood disorders that include
hemophilia A. Sernova is currently focused on developing a ‘functional cure’ for insulin-dependent diabetes
with its lead technology, the Cell Pouch System, a novel implantable and scalable medical device with immune
protected therapeutic cells.
On implantation, The Cell Pouch forms a natural, vascularized tissue environment in the body allowing long-
term survival and function of therapeutic cells that release essential factors that are absent or deficient in the
bodies of patients with certain chronic diseases. Sernova’s Cell Pouch System has demonstrated its potential to
be a ‘functional cure’ for people with T1D in an ongoing Phase 1/2 clinical study at the University of Chicago.
Sernova partnered with Evotec to develop an implantable off-the-shelf iPSC (induced pluripotent stem cells)
based islet replacement therapy. This partnership provides Sernova a potentially unlimited supply of insulin-
producing cells to treat millions of patients with insulin-dependent diabetes (type 1 and type 2). Sernova’s
development pipeline that uses its Cell Pouch System also includes: a cell therapy for hypothyroid disease
resulting from thyroid gland removal and an ex vivo lentiviral Factor VIII gene therapy for hemophilia A.
FOR FURTHER INFORMATION, PLEASE CONTACT:
Christopher Barnes
VP, Investor Relations
Sernova Corp.
Tel: +1 519-902-7923
Email: christopher.barnes@sernova.com
Website: www.sernova.com
FORWARD-LOOKING INFORMATION
This release contains statements that, to the extent they are not recitations of historical facts, may constitute
“forward-looking statements” that involve various risks, uncertainties, and assumptions, including, without
limitation, statements regarding the prospects, plans, and objectives of the company. Wherever possible, but
not always, words such as "expects", "plans", "anticipates", "believes", "intends", "estimates", "projects",
"potential for" and similar expressions, or that events or conditions "will", "would", "may", "could" or "should"
occur are used to identify forward-looking statements. These statements reflect management’s beliefs with
respect to future events and are based on information currently available to management on the date such
statements were made. Many factors could cause Sernova’s actual results, performances or achievements to
not be as anticipated, estimated or intended or to differ materially from those expressed or implied by the
forward-looking statements contained in this news release. Such factors could include, but are not limited to,
the company’s ability to secure additional financing and licensing arrangements on reasonable terms, or at all;
ability to conduct all required preclinical and clinical studies for the company’s Cell Pouch System and or
related technologies, including the timing and results of those trials; ability to obtain all necessary regulatory
approvals, or on a timely basis; ability to in-license additional complementary technologies; ability to execute
its business strategy and successfully compete in the market; and the inherent risks associated with the
development of biotechnology combination products generally. Many of the factors are beyond our control,
including those caused by, related to, or impacted by the novel coronavirus pandemic. Investors should consult
the company’s quarterly and annual filings available on www.sedarplus.ca for additional information on risks
and uncertainties relating to the forward-looking statements. Sernova expressly disclaims any intention or
obligation to update or revise any forward-looking statements, whether as a result of new information, future
events or otherwise.
Press Release Communiqué de presse - Avril 22, 2024 22 Avril, 2024
Sernova Welcomes Dr. Bernd Muehlenweg as Evotec’s Nominee for its Board of Directors
Dr. Daniel Mahony to step down to pursue new commitment
LONDON, Ontario; BOSTON, Massachusetts – April 22, 2024 – Sernova Corp. (TSX:SVA)
(OTCQB:SEOVF) (FSE/XETRA:PSH), a clinical-stage biotechnology company focused on the
development of regenerative medicine cell therapies for treatment of chronic diseases, announces
today that Dr. Bernd Muehlenweg, Senior Vice President of Global Business Development at Evotec
SE (Frankfurt Stock Exchange: EVT, MDAX/TecDAX, ISIN: DE0005664809; NASDAQ: EVO), has been
appointed to its Board of Directors. Evotec recently shared that due to the impending departure of Dr. Daniel
Mahony, its current Sernova Board delegate, who is leaving to pursue a new professional
undertaking, they would be nominating a new Board representative. With heartfelt gratitude, we
wish Dan all the very best in his future endeavors.
Dr. Muehlenweg will join Sernova’s Board of Directors effective immediately. Sernova expects the
brief period of overlap with Dr. Daniel Mahony will assist in the transition bringing Dr. Muehlenweg
rapidly up to speed on Sernova’s operational and strategic matters.
Bernd Muehlenweg's work experience includes various senior leadership positions in the biotech
and pharmaceutical industry with a concentration on business development and alliance
management roles. At Evotec, he leads the company's partnering and out-licensing efforts in their
focus areas including iPSC-based Cell Therapy, Panomics-driven drug discovery, Oncology,
Immunology & Inflammation, Infectious Diseases, Predictive Safety and Enabling Technologies.
Prior to joining Evotec, he held the position of Chief Business Officer and served on the Executive
Board of Nanobiotix, a French clinical stage oncology company. Bernd co-founded Panoptes
Pharma GmbH, an Austrian biotech company focused on developing therapies for eye diseases,
which was later acquired by Eyegate Pharmaceuticals. Additionally, at Wilex AG, he played a key role
in the company's growth and expansion. He began his career as a Group Leader at the Technical
University of Munich, Germany.
Bernd Muehlenweg graduated with a Ph.D. from the oncology research group at the Department of
Gynecology at the Technical University of Munich in 2000. He further attended management classes
at the Switzerland based St. Galler Business School in 2006.
“Sernova’s Cell Pouch and Evotec’s iPSC derived islet like clusters are a powerful combination,
offering a potential functional cure for type 1 diabetes (T1D) in the not-too-distant future. My
personal goal is to foster sustainable, fruitful and durable partnerships to advance scientific
innovations into approved treatments. I have tremendous confidence that the strategic
collaboration between Evotec and Sernova will provide a strong foundation to potentially achieve
this objective” said Bernd Muehlenweg, Sernova’s new Board Director.
“At Sernova, we will continue to pursue opportunities to improve the lives of patients with unmet
needs while building long-term value for shareholders in multiple ways. I cannot stress enough the
importance of the Cell Pouch System as the anchor for our ongoing T1D trial. In addition to its longterm
payload survival, containment and retrievability characteristics, we believe that the Cell Pouch
is a key differentiator in delivering clinically meaningful outcomes, including insulin independence
and normalized HbA1c counts amongst others, to several patients in our ongoing Phase 1/2 trial. We
are pleased to welcome Bernd as he joins us on our mission to build a future where chronic
conditions are no longer insurmountable obstacles.” said Cynthia Pussinen, CEO of Sernova.
ABOUT SERNOVA AND ITS CELL POUCH SYSTEM PLATFORM FOR CELL THERAPY
Sernova Corp. is a clinical-stage biotechnology company that is developing therapeutic cell
technologies for chronic diseases, including insulin-dependent diabetes, thyroid disease, and blood
disorders that include hemophilia A. Sernova is currently focused on developing a ‘functional cure’
for insulin-dependent diabetes with its lead technology, the Cell Pouch System, a novel implantable
and scalable medical device with immune protected therapeutic cells.
On implantation, The Cell Pouch forms a natural vascularized tissue environment in the body for
long-term survival and function of therapeutic cells that release essential factors that are absent or
deficient in the bodies of patients with certain chronic diseases. Sernova’s Cell Pouch System has
demonstrated its potential to be a ‘functional cure’ for people with T1D in an ongoing Phase 1/2
clinical study at the University of Chicago.
Sernova partnered with Evotec to develop an implantable off-the-shelf iPSC (induced pluripotent
stem cells) based islet replacement therapy. This partnership provides Sernova a potentially
unlimited supply of insulin-producing cells to treat millions of patients with insulin-dependent
diabetes (type 1 and type 2). Sernova’s development pipeline that uses its Cell Pouch System also
includes: a cell therapy for hypothyroid disease resulting from thyroid gland removal and an ex vivo
lentiviral Factor VIII gene therapy for hemophilia A.
FOR FURTHER INFORMATION, PLEASE CONTACT:
Christopher Barnes
VP, Investor Relations
Sernova Corp.
Tel: +1 519-902-7923
Email: christopher.barnes@sernova.com
Website: www.sernova.com
FORWARD-LOOKING INFORMATION
This release contains statements that, to the extent they are not recitations of historical facts, may
constitute “forward-looking statements” that involve various risks, uncertainties, and assumptions,
including, without limitation, statements regarding the prospects, plans, and objectives of the
company. Wherever possible, but not always, words such as "expects", "plans", "anticipates",
"believes", "intends", "estimates", "projects", "potential for" and similar expressions, or that events
or conditions "will", "would", "may", "could" or "should" occur are used to identify forward-looking
statements. These statements reflect management’s beliefs with respect to future events and are
based on information currently available to management on the date such statements were made.
Many factors could cause Sernova’s actual results, performances or achievements to not be as
anticipated, estimated or intended or to differ materially from those expressed or implied by the
forward-looking statements contained in this news release. Such factors could include, but are not
limited to, the company’s ability to secure additional financing and licensing arrangements on
reasonable terms, or at all; ability to conduct all required preclinical and clinical studies for the
company’s Cell Pouch System and or related technologies, including the timing and results of those
trials; ability to obtain all necessary regulatory approvals, or on a timely basis; ability to in-license
additional complementary technologies; ability to execute its business strategy and successfully
compete in the market; and the inherent risks associated with the development of biotechnology
combination products generally. Many of the factors are beyond our control, including those caused
by, related to, or impacted by the novel coronavirus pandemic. Investors should consult the
company’s quarterly and annual filings available on www.sedarplus.ca for additional information on
risks and uncertainties relating to the forward-looking statements. Sernova expressly disclaims any
intention or obligation to update or revise any forward-looking statements, whether as a result of
new information, future events or otherwise.
Press Release Communiqué de presse - Avril 02, 2024 2 Avril, 2024
Sernova Provides Positive Clinical and Platform Portfolio Update
Sernova Provides Positive Clinical and Platform Portfolio Update
LONDON, Ontario; BOSTON, Massachusetts – April 2, 2024 – Sernova Corp. (TSX:SVA) (OTCQB:SEOVF) (FSE/XETRA:PSH), a clinical-stage biotechnology company focused on the development of regenerative medicine cell therapies for treatment of chronic diseases, today provided a business update. Following a review of the company’s therapeutic pipeline and emerging opportunities for its Cell Pouch system platform technologies, Sernova confirms key priorities including its lighthouse program in insulin dependent Type 1 Diabetes plus its intention to advance an IND filing for its post-operative hypothyroidism program.
Data from a patient in Cohort 2 of the company’s lead clinical program for insulin dependent Type 1 diabetes (T1D) confirms histologic evidence of long-term (one year) robust survival of abundant human donor islets throughout the Cell Pouch. Additional Cohort 2 findings are specific to an advanced immunosuppression regimen planned for use in its upcoming Phase I/II trial with stem cell-derived islets under co-development with Evotec.
Cohort 2 patients treated with an advanced immunosuppression protocol avoided graft rejection and experienced minimal side eects in comparison to those patients observed in Cohort 1. None of the six patients in Cohort 2 treated with the advanced regimen have tested positive for donor specific antibodies (DSAs), a marker of graft rejection, in comparison to three of six patients who developed DSAs under the conventional immunosuppression regimen in Cohort 1. Ancillary medication, used in some Cohort 2 patients, demonstrated highly favorable graft survival and function for islets transplanted to the Cell Pouch and has been integrated into the updated regimen and implemented for all subsequent patient trial enrollments. The company anticipates reporting additional data from Cohort 2 of its ongoing Phase 1/2 clinical trial of its expanded 10-channel Cell Pouch during the second half of the year at major medical conferences. Sernova is pleased to report that this month marks the four-year anniversary of the first patient in Cohort 1 of this Phase 1/2 study who will celebrate insulin independence and normalized blood sugar levels, based on two transplants of human donor islets to the Cell Pouch plus a marginal portal vein top up.
“Based on the favorable results we are observing in ongoing pre-clinical studies, we have concluded that our hypothyroidism program represents another compelling opportunity by which to improve patients’ lives. We look forward to completing our pre-clinical work, engaging with regulatory agencies, and preparing for an IND filing later this year, with the goal of advancing a second indication into the clinic, further demonstrating the Cell Pouch as a drug delivery vehicle platform technology. Also of note, in addition to allowing for long term payload survival, our Cell Pouch has powerful containment and retrievability capabilities that we expect will have tremendous value for pharmaceutical companies looking to treat patients with cell therapies,” said Cynthia Pussinen, Chief Executive Oicer of Sernova.
“In parallel with these activities, and our ongoing hemophilia A work, we have identified several high value indications with unmet medical needs that could potentially benefit from our platform Cell Pouch technology, with an initial focus on endocrine disorders. In the coming months, we will be conducting commercial assessments to prioritize those areas where we can best extend our reach to more patients while creating enduring value for our shareholders. I am excited for what we are poised to achieve this year and look forward to providing further updates in the future,” Ms. Pussinen concluded.
ABOUT SERNOVA AND ITS CELL POUCH SYSTEM PLATFORM FOR CELL THERAPY
Sernova Corp. is a clinical-stage biotechnology company that is developing therapeutic cell technologies for chronic diseases, including insulin-dependent diabetes, thyroid disease, and blood disorders that include hemophilia A. Sernova is currently focused on developing a ‘functional cure’ for insulin-dependent diabetes with its lead technology, the Cell Pouch System, a novel implantable and scalable medical device with immune protected therapeutic cells. On implantation, The Cell Pouch forms a natural vascularized tissue environment in the body for long-term survival and function of therapeutic cells that release essential factors that are absent or deficient in the bodies of patients with certain chronic diseases. Sernova’s Cell Pouch System has demonstrated its potential to be a ‘functional cure’ for people with T1D in an ongoing Phase 1/2 clinical study at the University of Chicago.
Sernova partnered with Evotec to develop an implantable off-the-shelf iPSC (induced pluripotent stem cells) based islet replacement therapy. This partnership provides Sernova a potentially unlimited supply of insulin-producing cells to treat millions of patients with insulin-dependent diabetes (type 1 and type 2). Sernova’s development pipeline that uses its Cell Pouch System also includes: a cell therapy for hypothyroid disease resulting from thyroid gland removal and an ex vivo lentiviral Factor VIII gene therapy for hemophilia A.
FOR FURTHER INFORMATION, PLEASE CONTACT:
- Christopher Barnes
- VP, Investor Relations
- Sernova Corp.
- Tel: +1 519-902-7923
- Email: christopher.barnes@sernova.com
- Website: www.sernova.com
FORWARD-LOOKING INFORMATION
This release contains statements that, to the extent they are not recitations of historical facts, may constitute “forward-looking statements” that involve various risks, uncertainties, and assumptions, including, without limitation, statements regarding the prospects, plans, and objectives of the company. Wherever possible, but not always, words such as "expects", "plans", "anticipates", "believes", "intends", "estimates", "projects", "potential for" and similar expressions, or that events or conditions "will", "would", "may", "could" or "should" occur are used to identify forward-looking statements. These statements reflect management’s beliefs with respect to future events and are based on information currently available to management on the date such statements were made. Many factors could cause Sernova’s actual results, performances or achievements to not be as anticipated, estimated or intended or to dier materially from those expressed or implied by the forward-looking statements contained in this news release. Such factors could include, but are not limited to, the company’s ability to secure additional financing and licensing arrangements on reasonable terms, or at all; ability to conduct all required preclinical and clinical studies for the company’s Cell Pouch System and or related technologies, including the timing and results of those trials; ability to obtain all necessary regulatory approvals, or on a timely basis; ability to in-license additional complementary technologies; ability to execute its business strategy and successfully compete in the market; and the inherent risks associated with the development of biotechnology combination products generally. Many of the factors are beyond our control, including those caused by, related to, or impacted by the novel coronavirus pandemic. Investors should consult the company’s quarterly and annual filings available on www.sedarplus.ca for additional information on risks and uncertainties relating to the forward-looking statements. Sernova expressly disclaims any intention or obligation to update or revise any forward-looking statements, whether as a result of new information, future events or otherwise.
Press Release Communiqué de presse - Mars 11, 2024 11 Mars, 2024
Sernova Announces Management Developments
LONDON, Ontario; BOSTON, Massachusetts – March 11, 2024 – Sernova Corp. (TSX:SVA) (OTCQB:SEOVF)
(FSE/XETRA:PSH), a clinical-stage company focused on providing regenerative medicine therapeutics to
patients with chronic conditions, today announced that Nicholas J. Rossettos, CPA has joined Sernova on a
consulting basis as interim Chief Financial Officer (CFO).
With a wealth of experience in financial leadership within the biotech industry, Nick brings a fresh perspective
and a strong track record of success to the Sernova team during this transition period. The company has
initiated a formal search for a permanent CFO and is also actively recruiting additional talent to fill key
leadership roles and strengthen its senior executive team, which started with the additions of Cynthia Pussinen
as the Chief Executive Officer and Modestus Obochi as Chief Business Officer. The goal is to ensure that
Sernova has the expertise to execute its strategic vision and drive the company forward by delivering life-
changing therapies to patients worldwide.
David Swetlow, Chief Financial Officer, is no longer with the company. Mr. Swetlow’s employment was
terminated for cause after the Board received and considered findings made by independent legal counsel in
connection with an ongoing investigation into alleged misconduct. Another senior officer of the company has
been placed on administrative leave pending the final outcome of the investigation. None of the allegations, if
substantiated, are expected to materially change or impact the Company's financial statements or its reporting
obligations. The Sernova mission remains unchanged: to improve the lives of patients through groundbreaking
innovation and compassionate care. The board and the senior leadership team are deeply grateful for the
dedication and hard work of the Company’s employees, partners, and stakeholders, and remains steadfast in
its commitment to maintaining the highest standards of integrity, transparency, and accountability in all aspects
of our operations.
ABOUT SERNOVA CORP. AND THE CELL POUCH SYSTEM PLATFORM FOR CELL THERAPY
Sernova Corp. is a clinical-stage biotechnology company that is developing therapeutic cell technologies for
chronic diseases, including insulin-dependent diabetes, thyroid disease, and blood disorders that include
hemophilia A. Sernova is currently focused on developing a ‘functional cure’ for insulin-dependent diabetes
with its lead asset, the Cell Pouch System, a novel implantable and scalable medical device with immune
protected therapeutic cells. On implantation, The Cell Pouch forms a natural vascularized tissue environment in
the body for long-term survival and function of therapeutic cells that release essential factors that are absent or
deficient in the bodies of patients with certain chronic diseases. Sernova’s Cell Pouch System has demonstrated
its potential to be a ‘functional cure’ for people with T1D in an ongoing Phase 1/2 clinical study at the University
of Chicago. In May 2022, Sernova and Evotec entered into a global strategic partnership to develop an
implantable off-the-shelf iPSC (induced pluripotent stem cells) based islet replacement therapy. This
partnership provides Sernova a potentially unlimited supply of insulin-producing cells to treat millions of
patients with insulin-dependent diabetes (type 1 and type 2). Sernova continues to progress two additional
development programs that utilize its Cell Pouch System: a cell therapy for hypothyroid disease resulting from
thyroid gland removal and an ex vivo lentiviral Factor VIII gene therapy for hemophilia A.
FOR FURTHER INFORMATION, PLEASE CONTACT:
Christopher Barnes
VP, Investor Relations
Sernova Corp.
christopher.barnes@sernova.com
Tel: 519-902-7923
www.sernova.com
FORWARD-LOOKING INFORMATION
This release contains statements that, to the extent they are not recitations of historical facts, may constitute
“forward-looking statements” that involve various risks, uncertainties, and assumptions, including, without
limitation, statements regarding the prospects, plans, and objectives of the company. Wherever possible, but
not always, words such as "expects", "plans", "anticipates", "believes", "intends", "estimates", "projects",
"potential for" and similar expressions, or that events or conditions "will", "would", "may", "could" or "should"
occur are used to identify forward-looking statements. These statements reflect management’s beliefs with
respect to future events and are based on information currently available to management on the date such
statements were made. Many factors could cause Sernova’s actual results, performances or achievements to
not be as anticipated, estimated or intended or to differ materially from those expressed or implied by the
forward-looking statements contained in this news release. Such factors could include, but are not limited to,
the company’s ability to secure additional financing and licensing arrangements on reasonable terms, or at all;
ability to conduct all required preclinical and clinical studies for the company’s Cell Pouch System and or
related technologies, including the timing and results of those trials; ability to obtain all necessary regulatory
approvals, or on a timely basis; ability to in-license additional complementary technologies; ability to execute
its business strategy and successfully compete in the market; and the inherent risks associated with the
development of biotechnology combination products generally. Many of the factors are beyond our control,
including those caused by, related to, or impacted by the novel coronavirus pandemic. Investors should consult
the company’s quarterly and annual filings available on www.sedarplus.ca for additional information on risks
and uncertainties relating to the forward-looking statements. Sernova expressly disclaims any intention or
obligation to update or revise any forward-looking statements, whether as a result of new information, future
events or otherwise.
Press Release Communiqué de presse - Janvier 29, 2024 29 Janvier, 2024
Sernova Provides Recap of 2023 Accomplishments and Anticipated 2024 Milestones
- Recently completed recruitment of all 7 patients in Cohort 2
- Anticipates additional data from Cohort 2 of Phase 1/2 clinical trial evaluating its enhanced 10-channel
Cell Pouch in patients with type 1 diabetes (T1D) in Q1/24
- Expects to report preclinical data supporting an IND application to evaluate the use of therapeutic cells in
combination with Cell Pouch in patients with post-operative hypothyroidism
LONDON, Ontario; Boston, Massachusetts – January 29, 2024 – Sernova Corp. (TSX:SVA) (OTCQB:SEOVF)
(FSE/XETRA:PSH), a clinical-stage company and leader in cell therapeutics, today provided a business
update, including a recap of 2023 accomplishments and a preview of certain milestones anticipated in 2024.
“In 2023, we added to the compelling and expanding set of data demonstrating the safety and efficacy of our
novel cell therapy platform for chronic diseases, including our higher capacity 10-channel Cell Pouch that is
being evaluated in our ongoing Phase 1/2 human donor islet clinical trial in T1D,” said Cynthia Pussinen, Chief
Executive Officer of Sernova. “We also made significant progress with our hemophilia A program, including
receipt of both Orphan Drug and Rare Pediatric Disease designations from the U.S. Food and Drug
Administration (FDA). Lastly, we announced an exciting preclinical research collaboration with AstraZeneca
that has the potential to significantly expand the use of the Cell Pouch in additional high-value indications.”
“Looking ahead to 2024, we anticipate additional patient data from Cohort 2 of our T1D human donor islet
trial, as well as important data from our thyroid disease program as we work to further our preclinical pipeline.
We continue to advance plans for our next T1D Phase 1/2 clinical study utilizing Evotec’s iPSC-derived islet
like clusters (ILCs) in our Cell Pouch. Use of iPSCs provides significant advantages over human donor islets,
including the ability to scale this promising treatment to commercially viable levels enabling the treatment of
millions of patients. Throughout 2024 our strategic partner, Evotec, will continue to optimize and advance the
development of iPSC derived ILCs for use in additional IND enabling studies and clinical trials. Given the
complexity around scaling-up of iPSCs and therapeutic cell manufacturing, as well as the relative nascency of
the entire advanced therapeutics field, timelines have extended, and we now anticipate initiating a clinical trial
evaluating our Cell Pouch with iPSC-derived ILCs in the fourth quarter of 2025.” Ms. Pussinen added.
“I am encouraged with our progress and believe we have built a solid foundation consisting of a portfolio of
fundamentally transformational medical treatments for patients living with chronic conditions that will result in
multiple potentially value-creating milestones this year and next,” Ms. Pussinen concluded.
2023 Achievements:
• Announced senior leadership additions, including Cynthia Pussinen as new Chief Executive Officer, and
Dr. Modestus Obochi, Ph.D., MBA, as Chief Business Officer
• Received both Orphan Drug Designation (ODD) and Rare Pediatric Disease Designation (RPDD) from the
U.S. FDA for the Company’s hemophilia A program
• Announced positive interim data from Cohorts 1 and 2 of the ongoing Phase 1/2 human donor islet
clinical trial of its Cell Pouch System™ in patients with T1D at the 2023 IPITA-IXA-CTRMS Joint Congress.
Notably, five of the six patients in Cohort 1 were able to discontinue insulin therapy following islet
transplantation, and all six patients achieved HbA1c values in the non-diabetic range (<6.5%). In Cohort
2, the first six of seven planned patients received the higher capacity 10-channel Cell Pouch and five
patients received a first islet transplant. Stable fasting and stimulated serum C-peptide levels were
observed following a single islet transplant into the 10-channel Cell Pouch in the first assessable Cohort 2
patient who subsequently achieved insulin independence with a modest portal vein top-up.
• Announced positive results from its conformal coating immune protection technology program that is
used in combination with the Cell Pouch System™ and is intended to eliminate the need for chronic
immunosuppression medications.
• Presented preclinical data supporting the planned Phase 1/2 clinical trial of Evotec’s “off-the-shelf” iPSC-
derived ILCs in combination with Sernova’s Cell Pouch System for the treatment of patients with T1D.
Specifically, data demonstrated that Evotec’s iPSC-derived ILCs generated robust and durable insulin
independence with blood C-peptide levels and glucose tolerance test results equivalent to a test group
with human islets. A separate study showed sustained normalization of blood sugar levels in diabetic mice
throughout the 320-day term of the study. Human testing is anticipated to begin in late 2025.
• Announced a research collaboration with AstraZeneca to evaluate the use of Sernova’s Cell Pouch System
in combination with AstraZeneca’s novel therapeutic cells for various indications. The discovery work is
being funded and conducted by AstraZeneca.
• Completed recruitment of all 7 patients in Cohort 2 of the ongoing Phase 1/2 trial using the 10-channel
Cell Pouch.
Anticipated Select 2024 Milestones:
• Additional data from Cohort 2 of the ongoing U.S. Phase 1/2 clinical trial, which is evaluating its enhanced
10-channel Cell Pouch in patients with T1D, are expected beginning in Q1.
• Additional preclinical data to support an IND application to evaluate the Company’s autograft thyroid
cells in patients suffering from thyroid disease, with no immunosuppressive regimen required.
• Completion of thyroid disease market study validating the current market size and detailing the unmet
medical need.
• Potential engagement with additional life sciences focused companies, academic institutions and external
parties through partnership and collaboration opportunities, which could be announced over the course
of 2024.
• Additional funding to support growth through strategic alliances, credit facilities and/or institutional
equity financings with the goal of maximizing shareholder value.
Fiscal Year 2023 Financials
Today, Sernova filed its financial results on SEDAR for the fiscal year 2023.
ABOUT SERNOVA CORP. AND THE CELL POUCH SYSTEM PLATFORM FOR CELL THERAPY
Sernova Corp. is a clinical-stage biotechnology company that is developing therapeutic cell technologies for
chronic diseases, including insulin-dependent diabetes, thyroid disease, and blood disorders that include
hemophilia A. Sernova is currently focused on developing a ‘functional cure’ for insulin-dependent diabetes
with its lead asset, the Cell Pouch System, a novel implantable and scalable medical device with immune
protected therapeutic cells. On implantation, The Cell Pouch forms a natural vascularized tissue environment
in the body for long-term survival and function of therapeutic cells that release essential factors that are
absent or deficient in the bodies of patients with certain chronic diseases. Sernova’s Cell Pouch System has
demonstrated its potential to be a ‘functional cure’ for people with T1D in an ongoing Phase 1/2 clinical study
at the University of Chicago. Sernova is also advancing a proprietary technology in collaboration with the
University of Miami to shield therapeutic cells from immune system attack with the goal to eliminate the need
for chronic, systemic immunosuppression. In May 2022, Sernova and Evotec entered into a global strategic
partnership to develop an implantable off-the-shelf iPSC (induced pluripotent stem cells) based islet
replacement therapy. This partnership provides Sernova a potentially unlimited supply of insulin-producing
cells to treat millions of patients with insulin-dependent diabetes (type 1 and type 2). Sernova continues to
progress two additional development programs that utilize its Cell Pouch System: a cell therapy for
hypothyroid disease resulting from thyroid gland removal and an ex vivo lentiviral Factor VIII gene therapy for
hemophilia A.
FOR FURTHER INFORMATION, PLEASE CONTACT:
Christopher Barnes
VP, Investor Relations
Sernova Corp.
christopher.barnes@sernova.com
Tel: 519-902-7923
www.sernova.com
FORWARD-LOOKING INFORMATION
This release contains statements that, to the extent they are not recitations of historical facts, may constitute
“forward-looking statements” that involve various risks, uncertainties, and assumptions, including, without
limitation, statements regarding the prospects, plans, and objectives of the company. Wherever possible, but
not always, words such as "expects", "plans", "anticipates", "believes", "intends", "estimates", "projects",
"potential for" and similar expressions, or that events or conditions "will", "would", "may", "could" or
"should" occur are used to identify forward-looking statements. Forward looking statements in this news
release include, without limitation, statements relating to the timing of clinical trials, the timing with respect to
the receipt of data from on-going clinical trials, timing with respect to preclinical data, the completion of
thyroid disease market study, statements with respect to the potential future engagement with additional
universities, life sciences focus companies and external parties and the timing thereof.
These statements reflect the current expectations, assumptions and beliefs of management currently available
to it on the date such statements were made, including Sernova’s ability to secure additional financing and
licensing arrangements; the timing with respect to the engineering and scaling-up of Sernova’s technologies;
the ability to conduct all required preclinical and clinical studies for the company’s Cell Pouch System and or
related technologies; the timing and results of preclinical and clinical trials; the ability to obtain all necessary
regulatory approvals on a timely basis; the ability to in-license additional complementary technologies; and
the ability of Sernova to execute its business strategy, attract additional partners and successfully compete in
the market.
Although the Company believes that the assumptions inherent in these forward-looking statements are
reasonable, forward-looking statements are not a guarantee of future performance and accordingly undue
reliance should not be placed on such statements. Forward-looking statements are subject to a number of
risks and uncertainties, some that are similar to biotechnology companies and some that are unique to
Sernova. The actual results may differ materially from those expressed or implied by the forward-looking
information, and even if such actual results are realized or substantially realized, there can be no assurance
that they will have the expected consequences to, or effects on, Sernova. Sernova’s annual information form
dated January 29, 2024, its annual management's discussion and analysis for the year ended October 31,
2023, and other documents it files from time to time with securities regulatory authorities describe the risks,
uncertainties, material assumptions and other factors that could influence actual results and such factors are
incorporated herein by reference. Copies of these documents are available without charge by referring to the
company's profile on SEDAR+ at www.sedarplus.ca. All forward-looking statements speak only as of the date
on which they were made and, except as may be required by applicable securities laws, the Company
disclaims any intent or obligation to update any forward-looking statements, whether as a result of new
information, future events or results or otherwise.
Press Release Communiqué de presse - Novembre 27, 2023 27 Novembre, 2023
Sernova Receives Orphan Drug and Rare Pediatric Disease Designations for its Hemophilia A Program from FDA
Sernova announces research collaboration with Professor Antonia Follenzi, expert in hemophilia A at the
University of Piemonte Orientale
LONDON, Ontario; WINDHAM COUNTY, Connecticut – November 27, 2023 – Sernova Corp. (TSX:SVA)
(OTCQB:SEOVF) (FSE/XETRA:PSH), a clinical-stage company and leader in cell therapeutics, today
announced the U.S. Food and Drug Administration (FDA) has granted both Orphan Drug Designation (ODD) and
Rare Pediatric Disease Designation (RPDD) for the company’s Hemophilia A program.
The FDA grants orphan designation, also referred to as orphan status, to therapies intended for the treatment
of rare diseases that affect fewer than 200,000 people in the US. This designation provides certain benefits,
including tax credits for qualified clinical testing, waiver or partial payment of FDA application fees and seven
years of market exclusivity, if approved. Separately, rare pediatric disease designations are granted for rare
diseases that primarily affect children under 18 years old with recipients of this designation being awarded a
priority review voucher, upon approval. The priority review voucher may be redeemed, transferred, or sold.
“We are pleased with the FDA’s decision to grant these designations for our novel treatment for Hemophilia A,
which uses the Cell Pouch™ in combination with cells corrected for the production of Factor VIII,” commented
Cynthia Pussinen, Chief Executive Officer of Sernova. “Hemophilia A is a serious, life limiting condition and we
are committed to advancing development of the program, with a hope to positively impact patients around the
world who are waiting for improved treatments.”
About Sernova’s Hemophilia A Cell Pouch System Program
Sernova’s Hemophilia A program combines the Sernova Cell Pouch™ with a patient’s own cells and will not
require the use of immunosuppression medications. This therapy is intended to replace Factor VIII (FVIII) - an
essential blood-clotting protein that is deficient or absent in patients with hemophilia A; this is accomplished by
correcting the patient’s own Blood Outgrowth Endothelial Cells (BOECs) and subsequently returning them to
the patient via the Cell Pouch™. These modified cells function to release FVIII into the bloodstream, restoring
the patient’s ability for clotting during periods of bleeding.
Sernova and research partners, through a Horizon 2020 grant which is part of the EU's research and innovation
funding program in proof-of-concept work, successfully corrected human blood cells from patients with
hemophilia A to produce Factor VIII using a novel first-in-class gene and cell therapy approach where the
corrected cells were transplanted into the pre-implanted, vascularized Cell Pouch in a preclinical model of
Hemophilia A. The work demonstrated an improvement in blood clotting using the combined technologies
(Efficient and safe correction of hemophilia A by lentiviral vector-transduced BOECs in an implantable device;
Molecular Therapy: Methods & Clinical Development Vol.23, Dec 2021).
Collaboration with the University of Piemonte Orientale
The company is pleased to announce a collaboration with the University of Piemonte Orientale, Italy under the
direction of Antonia Follenzi MD, Ph.D. Professor of Histology and Cell and Gene therapy. Dr. Follenzi is a
pioneer of cell and gene therapy approaches to cure Hemophilia A. Her laboratory has expertise in the
generation of BOECs from Hemophilic patients and correcting the FVIII gene using Lentiviral vectors.
The purpose of the new collaboration is to optimize the technology using lentiviral vectors to drive the
expression of FVIII transgene under the control of novel promoters into BOECs of hemophilia patients to
achieve optimal sustained production of FVIII using an optimized cell dose within the Cell Pouch in an animal
model of Hemophilia A. The overall goal of the collaboration is to develop a product combination along with
pre-clinical results that support advancing into clinical trials in patients with Hemophilia A.
Professor Follenzi stated, “I am pleased to be working with the Sernova team on these advanced technologies
for a new and safe treatment of patients with Hemophilia A using a gene and cell therapy approach within the
Cell Pouch. Our goal is to advance these new technologies to further maximize the release of FVIII into the
bloodstream and to extend the duration of that release for a sufficient duration of time to eliminate the need
for weekly infusions of FVIII and to significantly improve the lives of people with Hemophilia A.”
BENEFITS OF ODD AND RPDD
Combined benefits of these designations include exclusive marketing rights for a seven-year period, after
marketing approval, a 25% federal tax credit for clinical research expenses incurred in the US which is
applicable for up to 20 years, waiver of Prescription Drug User Fee Act (PDUFA) fees for orphan drugs
(currently worth >US$3 million), ability to qualify to compete for research grants from the Office of Orphan
Products Development (OOPD) to support clinical studies for the orphan indication, and eligibility to receive
regulatory assistance and guidance from FDA to design the development plan.
Furthermore, once the therapy is approved for marketing, it cannot be copied and sold in the US for 7 years
regardless of patent life and the sponsor will be granted a Priority Voucher which can be used to receive
approximately 4 months reduction time of the standard FDA review period or sold.
ABOUT HEMOPHILIA A
Hemophilia encompasses a group of inherited disorders that alter blood coagulation. Classical hemophilia,
also known as hemophilia A, is a hereditary hemorrhagic disorder resulting from a congenital deficit of FVIII
that manifests as protracted and excessive bleeding either spontaneously or secondary to trauma. 1
Hemophilia A is the most common form of Hemophilia and is a genetic disorder caused by missing or defective
FVIII, a blood clotting protein. Severe Hemophilia A occurs in about 60% of cases where the deficiency of FVIII
is less than 1% of normal blood concentration. While it is passed down from parents to children, about 1/3 of
cases are caused by a spontaneous change in the gene.
According to the US Centers for Disease Control and Prevention, Hemophilia A occurs in about 1 in 5,000
births. Prolonged bleeding, in areas such as the brain, of a person with Hemophilia A, can be fatal. Prolonged
bleeding in joints can cause inflammatory responses and permanent joint damage. Approximately 20,000
people in the United States, 2,500 in Canada and 10,000 in Europe have moderate to severe forms of
Hemophilia A. Though there is no cure for the disease, Hemophilia A can be controlled with regular infusions of
recombinant clotting FVIII. Treatment costs per patient are as high as US $200,000 or more each year, with an
aggregate therapeutic cost of over US$ 10 billion per year.
ABOUT SERNOVA CORP. AND THE CELL POUCH SYSTEM PLATFORM FOR CELL THERAPY
Sernova Corp. is a clinical-stage biotechnology company that is developing therapeutic cell technologies for
chronic diseases, including insulin-dependent diabetes, thyroid disease, and blood disorders that include
hemophilia A. Sernova is currently focused on developing a ‘functional cure’ for insulin-dependent diabetes
with its lead asset, the Cell Pouch System, a novel implantable and scalable medical device with immune
protected therapeutic cells. On implantation, The Cell Pouch forms a natural vascularized tissue environment
in the body for long-term survival and function of therapeutic cells that release essential factors that are
absent or deficient in the bodies of patients with certain chronic diseases. Sernova’s Cell Pouch System has
demonstrated its potential to be a ‘functional cure’ for people with T1D in an ongoing Phase 1/2 clinical study
at the University of Chicago. Sernova is also advancing a proprietary technology in collaboration with the
University of Miami to shield therapeutic cells from immune system attack with the goal to eliminate the need
for chronic, systemic immunosuppression. In May 2022, Sernova and Evotec entered into a global strategic
partnership to develop an implantable off-the-shelf iPSC (induced pluripotent stem cells) based islet
replacement therapy. This partnership provides Sernova a potentially unlimited supply of insulin-producing
cells to treat millions of patients with insulin-dependent diabetes (type 1 and type 2). Sernova continues to
progress two additional development programs that utilize its Cell Pouch System: a cell therapy for
hypothyroid disease resulting from thyroid gland removal and an ex vivo lentiviral Factor VIII gene therapy for
hemophilia A.
FOR FURTHER INFORMATION, PLEASE CONTACT:
Corporate:
Christopher Barnes
VP, Investor Relations
Sernova Corp.
christopher.barnes@sernova.com
Tel: 519-902-7923
www.sernova.com
Investors:
Corey Davis, Ph.D.
LifeSci Advisors, LLC
cdavis@lifesciadvisors.com
Tel: 212-915-2577
Media:
Hannah Holmquist
LifeSci Communications
hholmquist@lifescicomms.com
Tel: 619-723-4326
FORWARD-LOOKING INFORMATION
This release contains statements that, to the extent they are not recitations of historical facts, may constitute
“forward-looking statements” that involve various risks, uncertainties, and assumptions, including, without
limitation, statements regarding the prospects, plans, and objectives of the company. Wherever possible, but
not always, words such as "expects", "plans", "anticipates", "believes", "intends", "estimates", "projects",
"potential for" and similar expressions, or that events or conditions "will", "would", "may", "could" or
"should" occur are used to identify forward-looking statements. These statements reflect management’s
beliefs with respect to future events and are based on information currently available to management on the
date such statements were made. Many factors could cause Sernova’s actual results, performances or
achievements to not be as anticipated, estimated or intended or to differ materially from those expressed or
implied by the forward-looking statements contained in this news release. Such factors could include, but are
not limited to, the company’s ability to secure additional financing and licensing arrangements on reasonable
terms, or at all; ability to conduct all required preclinical and clinical studies for the company’s Cell Pouch
System and or related technologies, including the timing and results of those trials; ability to obtain all
necessary regulatory approvals, or on a timely basis; ability to in-license additional complementary
technologies; ability to execute its business strategy and successfully compete in the market; and the inherent
risks associated with the development of biotechnology combination products generally. Many of the factors
are beyond our control, including those caused by, related to, or impacted by the novel coronavirus pandemic.
Investors should consult the company’s quarterly and annual filings available on www.sedarplus.ca for
additional information on risks and uncertainties relating to the forward-looking statements. Sernova expressly
disclaims any intention or obligation to update or revise any forward-looking statements, whether as a result of
new information, future events or otherwise.
Press Release Communiqué de presse - Octobre 30, 2023 30 Octobre, 2023
Sernova Announces New Advancements of Conformal Coating Technology in Combination with the Cell Pouch System™ at the 2023 IPITA-IXA-CTRMS Joint Congress
LONDON, Ontario; WINDHAM COUNTY, Connecticut – October 30, 2023 – Sernova Corp. (TSX:SVA)
(OTCQB:SEOVF) (FSE/XETRA:PSH), a clinical-stage company and leader in cell therapeutics, today
announced an update on its conformal coating immune protection technology program, that is used in
combination with the Cell Pouch™.
The goal of the program is to eliminate the need for chronic immunosuppression medications, currently used in
islet cell transplantation for type 1 diabetes (T1D) treatment.
The update was presented by Dr. Alice Tomei, of the University of Miami, at the International Pancreas and
Islet Transplant Association (IPITA)-International Xenotransplantation Association (IXA)-Cell Transplant and
Regenerative Medicine Society (CTRMS) Joint Congress, taking place from October 26-29 in San Diego,
California.
The presentation showcased positive advancement in the refinement of the coating composition and process,
outcomes of preclinical studies with the Cell Pouch as well as rationale and historical development of the
technology.
The following summarizes key advancements presented:
• The final conformal coating composition exhibits significantly improved cell compatibility and overall
biocompatibility, representing evolution across years of process development work and preclinical testing.
• Coating process enhancements resulted in a 500% increase in conformal coating production capacity
(number of starting islets to be coated) and an 89% overall islet encapsulation yield (ratio of conformal
coated islets to initial islets). These enhancements have a direct positive impact on the in vitro and in vivo
efficacy of the coated islets.
• The final conformal coated product was purified using a process to contain 98% conformal coated islets
and only 2% empty capsules. This enables an increase in the number of functional coated islets that are
transplanted within the Cell Pouch chambers and minimization of graft volume.
• Using these composition and process development improvements, the coated islets were tested, in
combination with the Cell Pouch, in a syngeneic gold standard animal model of T1D to assess the safety
and efficacy of the combined product.
o Biocompatibility of the coated islets within the Cell Pouch was confirmed histologically demonstrating
healthy islets within the vascularized tissue matrix;
o Importantly, normal physiological transfer of glucose-stimulated insulin from the conformal coated islets
within the Cell Pouch was confirmed;
o Diabetic animals that received conformal coated islets within the Cell Pouch exhibited controlled blood
glucose to non-diabetic levels - which reversed upon removal of the Cell Pouch - proving function of the
conformal coated islets.
• A series of pilot studies using conformal coated islets, in combination with the Cell Pouch, in an allogeneic
rat model of T1D established the optimal conditions to achieve diabetes reversal. These conditions, which
are being used in confirmatory allogeneic studies in additional upcoming preclinical work, included:
o Drug kinetic studies which identified the optimal dose and frequency of a single selective immune
response agent to be used in combination with conformal coated islets;
o Islet dose-dependent glucose control was demonstrated using conformal coated islets in the Cell Pouch
with the selective immune response agent.
• The release criteria essential for clinical manufacturing have been developed, including coating
conformality, completeness, stiffness, thickness, and selective permeability. Using these criteria, the
conformal coating material showed long term mechanical stability, durability and selective permeability to
insulin and glucose molecules but not to antibodies or inflammatory cells. These are key requirements for
long-term function of the conformal coating technology in vivo.
• Significant progress was achieved in manufacturing of the coating scale up equipment. Prototype devices
are manufactured, and testing is in progress. Final system design will provide fully automated, GMP-
compliant coating applied to transplantable coated islets. The system function will involve conformal
coating, washing, counting and production monitoring.
“We have made significant advancements in the evolution of the conformal coating technology in our
collaboration with Sernova and are confident we have the final formulation. I look forward to our continuing
studies and presenting more results,” said Dr. Alice Tomei, Miami Engineering Career Development Associate
Professor of Biomedical Engineering at the University of Miami and director of the Islet Immunoengineering Lab
at the Miller School of Medicine Diabetes Research Institute.
“We understand that eliminating the standard immune suppression regimen is key to Sernova delivering a
‘functional cure’ for all T1D patients,” said Cynthia Pussinen, Chief Executive Officer at Sernova. “Our goal is to
continue to drive this technology ahead and ultimately bring a local immune-protected cell product into the
clinic combined with the Sernova Cell Pouch and iPSC islet-like clusters.”
RELEVANT PRESENTATION DETAILS
Title: Transplantation of Conformal Coated Islets in a Pre-Vascularized Cell Pouch™ Device for Beta Cell
Replacement in Diabetic Rats
Session: Islet Transplantation: Engineering the Islet Site Session
Date: Friday, October 27, 2023
ABOUT THE CONFORMAL COATING TECHNOLOGY
The conformal coating technology is a proprietary, mechanically stable, thin, biocompatible, and cross-linked
polymer hydrogel that coats the outside of each individual islet housed within the Cell Pouch. The conformal
coating technology offers selective permeability, with immuno-protective capability, while not interfering with
the kinetics of insulin and glucose release. Importantly, the conformal coating configuration also allows for the
physiological transfer of oxygen and nutrients needed for islet survival and long-term function within the pre-
vascularized Cell Pouch.
ABOUT SERNOVA CORP. AND THE CELL POUCH SYSTEM PLATFORM FOR CELL THERAPY
Sernova Corp. is a clinical-stage biotechnology company that is developing therapeutic cell technologies for
chronic diseases, including insulin-dependent diabetes, thyroid disease, and blood disorders that include
hemophilia A. Sernova is currently focused on developing a ‘functional cure’ for insulin-dependent diabetes
with its lead asset, the Cell Pouch System, a novel implantable and scalable medical device with immune
protected therapeutic cells. On implantation, The Cell Pouch forms a natural vascularized tissue environment
in the body for long-term survival and function of therapeutic cells that release essential factors that are
absent or deficient in the bodies of patients with certain chronic diseases. Sernova’s Cell Pouch System has
demonstrated its potential to be a ‘functional cure’ for people with T1D in an ongoing Phase 1/2 clinical study
at the University of Chicago. Sernova is also advancing a proprietary technology in collaboration with the
University of Miami to shield therapeutic cells from immune system attack with the goal to eliminate the need
for chronic, systemic immunosuppression. In May 2022, Sernova and Evotec entered into a global strategic
partnership to develop an implantable off-the-shelf iPSC (induced pluripotent stem cells) based islet
replacement therapy. This partnership provides Sernova a potentially unlimited supply of insulin-producing
cells to treat millions of patients with insulin-dependent diabetes (type 1 and type 2). Sernova continues to
progress two additional development programs that utilize its Cell Pouch System: a cell therapy for
hypothyroid disease resulting from thyroid gland removal and an ex vivo lentiviral Factor VIII gene therapy for
hemophilia A.
FOR FURTHER INFORMATION, PLEASE CONTACT:
Corporate:
Christopher Barnes
VP, Investor Relations
Sernova Corp.
christopher.barnes@sernova.com
Tel: 519-902-7923
www.sernova.com
Investors:
Corey Davis, Ph.D.
LifeSci Advisors, LLC
cdavis@lifesciadvisors.com
Tel: 212-915-2577
Media:
Hannah Holmquist
LifeSci Communications
hholmquist@lifescicomms.com
Tel: 619-723-4326
FORWARD-LOOKING INFORMATION
This release contains statements that, to the extent they are not recitations of historical facts, may constitute
“forward-looking statements” that involve various risks, uncertainties, and assumptions, including, without
limitation, statements regarding the prospects, plans, and objectives of the company. Wherever possible, but
not always, words such as "expects", "plans", "anticipates", "believes", "intends", "estimates", "projects",
"potential for" and similar expressions, or that events or conditions "will", "would", "may", "could" or
"should" occur are used to identify forward-looking statements. These statements reflect management’s
beliefs with respect to future events and are based on information currently available to management on the
date such statements were made. Many factors could cause Sernova’s actual results, performances or
achievements to not be as anticipated, estimated or intended or to differ materially from those expressed or
implied by the forward-looking statements contained in this news release. Such factors could include, but are
not limited to, the company’s ability to secure additional financing and licensing arrangements on reasonable
terms, or at all; ability to conduct all required preclinical and clinical studies for the company’s Cell Pouch
System and or related technologies, including the timing and results of those trials; ability to obtain all
necessary regulatory approvals, or on a timely basis; ability to in-license additional complementary
technologies; ability to execute its business strategy and successfully compete in the market; and the inherent
risks associated with the development of biotechnology combination products generally. Many of the factors
are beyond our control, including those caused by, related to, or impacted by the novel coronavirus pandemic.
Investors should consult the company’s quarterly and annual filings available on www.sedarplus.ca for
additional information on risks and uncertainties relating to the forward-looking statements. Sernova expressly
disclaims any intention or obligation to update or revise any forward-looking statements, whether as a result of
new information, future events or otherwise.
Press Release Communiqué de presse - Octobre 27, 2023 27 Octobre, 2023
Sernova Announces Positive Ongoing Interim Phase 1/2 Clinical Data for the Cell Pouch System™ for Type 1 Diabetes Trial at the 2023 IPITA, IXA, and CTRMS Joint Congress
• All six patients in the first cohort (Cohort A) were successfully implanted with the 8-channel Cell Pouch
System with post-transplant follow-up periods ranging from 6 months to 3.5 years;
• 5 of 6 patients in Cohort A discontinued insulin therapy (insulin independent) following islet
transplantation into the Cell Pouch and modest islet top-up via portal vein. All 6 patients achieved HbA1c
values in the non-diabetic range (<6.5%);
• In Cohort B, the first 6 of 7 planned patients have received the higher capacity 10-channel Cell Pouch and
5 patients have received a first islet transplant;
• Stable fasting and stimulated serum C-peptide levels were observed following a single islet transplant into
the 10-channel Cell Pouch in the first assessable Cohort B patient who subsequently achieved insulin
independence with a modest portal vein top up.
LONDON, Ontario; WINDHAM COUNTY, Connecticut – October 27, 2023 – Sernova Corp. (TSX:SVA)
(OTCQB:SEOVF) (FSE/XETRA:PSH), a clinical-stage company and leader in cell therapeutics, today presented
interim positive results from its ongoing Phase 1/2 clinical trial investigating islet allotransplantation into pre-
vascularized Sernova Cell Pouch™ during an oral presentation at the 2023 IPITA, IXA, and CTRMS Joint
Congress in San Diego, California.
Enrollment in Cohort A, which utilizes the 8-channel Cell Pouch, is complete with post-transplant data
available for periods of follow-up ranging from 6 months to 3.5 years. Enrollment in Cohort B, which utilizes
the higher capacity Cell Pouch and a revised and better-tolerated immunosuppressive regimen, began in
November 2022 and 6 of the 7 planned patients have now been successfully implanted.
The primary objective of the study is to investigate the safety and tolerability of islet transplantation into Cell
Pouch in patients with T1D, impaired hypoglycemia awareness, and a history of severe hypoglycemic episodes.
Secondary study objectives include establishment of islet release criteria predictive of outcomes from islet
transplant into the Cell Pouch and optimal dose and concentration ranges for purified islets transplanted into
the Cell Pouch.
Interim results from Cohort A demonstrated successful implantations of the 8-channel Cell Pouch in the 6
treated patients that were well tolerated with no seromas and no unexpected AEs (adverse events), chronic
pain or discomfort. Data showed histological evidence of surviving and functional islets and positive fasting
and stimulated serum C-peptide (a measure of islet insulin secretion) in patients who maintained optimal
immunosuppression. All 6 patients eventually received supplemental, marginal-dose islet infusions via the
portal vein with the first 5 having achieved sustained insulin independence. All 6 Cohort A patients achieved
HbA1c values in the non-diabetic range (<6.5%) with persistent serum fasting and stimulated C-peptide levels
for current durations out to 3.5 years.
In Cohort B, 6 of the planned 7 patients have been implanted with the higher capacity 10-channel Cell Pouch,
without complications. Among the 6 patients that have been implanted, 5 have completed at least one of the
two protocol-defined islet transplants to Cell Pouch.
The first assessable patient in Cohort B following the first Cell Pouch islet transplant showed persistent fasting
and stimulated serum C-peptide, with stable BETA-2 scores (a measure of islet graft function) that continued
at Day 180 following their first islet transplant to Cell Pouch. The same patient showed modest but favorable
improvements in HbA1c from 7.5% at baseline to 6.9% also at Day 180.
The day following the second islet transplant to Cell Pouch, results from a sample of the islets taken from the
donor pancreas on the day of transplant came back positive for the yeast, Candida albicans. Out of an
abundance of caution, Cell Pouches containing the contaminated islets were immediately removed. The Cell
Pouches that were previously transplanted with the first dose of uncontaminated, healthy islets were not
removed and remained in place, continuing to function. Explantation of the Cell Pouches containing
contaminated islets was completed without complications and the patient fully recovered without any wound
or systemic blood infection, demonstrating the designed retrievability of the transplanted Cell Pouch.
Following recovery, this patient received a modest intraportal islet transplant and remains insulin independent.
The revised immunosuppression protocol, used in Cohort B, continues to demonstrate favorable protection for
the islet grafts with no donor islet rejection or donor specific antibodies observed under the new regimen.
“I am pleased with the overall patient outcomes and learnings from the first trial cohort. We have applied those
learnings to the second patient cohort along with the introduction of the higher capacity 10-channel Cell
Pouch.” commented Dr. Piotr Witkowski, Director of the Pancreatic and Islet Transplant Program at The
University of Chicago, and principal investigator for the Sernova trial. “I am encouraged by the positive safety
profile observed with Cell Pouch implants longer than 4 years, and early patient outcomes with the enhanced
10-channel device that we are using in the second cohort. Enrollment of the second cohort is nearly complete,
and I look forward to reporting further results.”
“We are very encouraged by the results and our learnings from our trial to date.” said Cynthia Pussinen, Chief
Executive Officer at Sernova. “Having recently advanced the trial into Cohort B, using our higher capacity 10-
channel Cell Pouch, we are already seeing positive signals for both safety and efficacy. We look forward to
sharing the next trial update, in the coming months.”
These results were presented by Piotr Witkowski, Professor of Surgery at the University of Chicago at the
International Pancreas and Islet Transplant Association (IPITA), the International Xenotransplantation
Association (IXA), and the Cell Transplant and Regenerative Medicine Society (CTRMS) Joint Congress, taking
place from October 26 – 29 in San Diego, CA as an oral presentation entitled “Islet allotransplantation into pre-
vascularized Sernova Cell Pouch - Lessons learned from the first patient cohort” (Abstract #105, Session: “Islet
Transplantation: Engineering the Islet Site Session,” Thursday, October 26, 2023 2:45 p.m. to 3:45 p.m. PT)
For more information on the ongoing clinical study, go to clinicaltrials.gov (NCT03513939).
ABOUT SERNOVA CORP. AND THE CELL POUCH SYSTEM PLATFORM FOR CELL THERAPY
Sernova Corp. is a clinical-stage biotechnology company that is developing therapeutic cell technologies for
chronic diseases, including insulin-dependent diabetes, thyroid disease, and blood disorders that include
hemophilia A. Sernova is currently focused on developing a ‘functional cure’ for insulin-dependent diabetes
with its lead asset, the Cell Pouch System, a novel implantable and scalable medical device with immune
protected therapeutic cells. On implantation, The Cell Pouch forms a natural vascularized tissue environment
in the body for long-term survival and function of therapeutic cells that release essential factors that are
absent or deficient in the bodies of patients with certain chronic diseases. Sernova’s Cell Pouch System has
demonstrated its potential to be a ‘functional cure’ for people with T1D in an ongoing Phase 1/2 clinical study
at the University of Chicago. Sernova is also advancing a proprietary technology in collaboration with the
University of Miami to shield therapeutic cells from immune system attack with the goal to eliminate the need
for chronic, systemic immunosuppression. In May 2022, Sernova and Evotec entered into a global strategic
partnership to develop an implantable off-the-shelf iPSC (induced pluripotent stem cells) based islet
replacement therapy. This partnership provides Sernova a potentially unlimited supply of insulin-producing
cells to treat millions of patients with insulin-dependent diabetes (type 1 and type 2). Sernova continues to
progress two additional development programs that utilize its Cell Pouch System: a cell therapy for
hypothyroid disease resulting from thyroid gland removal and an ex vivo lentiviral Factor VIII gene therapy for
hemophilia A.
FOR FURTHER INFORMATION, PLEASE CONTACT:
Corporate:
Christopher Barnes
VP, Investor Relations
Sernova Corp.
christopher.barnes@sernova.com
Tel: 519-902-7923
www.sernova.com
Investors:
Corey Davis, Ph.D.
LifeSci Advisors, LLC
cdavis@lifesciadvisors.com
Tel: 212-915-2577
Media:
Hannah Holmquist
LifeSci Communications
hholmquist@lifescicomms.com
Tel: 619-723-4326
FORWARD-LOOKING INFORMATION
This release contains statements that, to the extent they are not recitations of historical facts, may constitute
“forward-looking statements” that involve various risks, uncertainties, and assumptions, including, without
limitation, statements regarding the prospects, plans, and objectives of the company. Wherever possible, but
not always, words such as "expects", "plans", "anticipates", "believes", "intends", "estimates", "projects",
"potential for" and similar expressions, or that events or conditions "will", "would", "may", "could" or
"should" occur are used to identify forward-looking statements. These statements reflect management’s
beliefs with respect to future events and are based on information currently available to management on the
date such statements were made. Many factors could cause Sernova’s actual results, performances or
achievements to not be as anticipated, estimated or intended or to differ materially from those expressed or
implied by the forward-looking statements contained in this news release. Such factors could include, but are
not limited to, the company’s ability to secure additional financing and licensing arrangements on reasonable
terms, or at all; ability to conduct all required preclinical and clinical studies for the company’s Cell Pouch
System and or related technologies, including the timing and results of those trials; ability to obtain all
necessary regulatory approvals, or on a timely basis; ability to in-license additional complementary
technologies; ability to execute its business strategy and successfully compete in the market; and the inherent
risks associated with the development of biotechnology combination products generally. Many of the factors
are beyond our control, including those caused by, related to, or impacted by the novel coronavirus pandemic.
Investors should consult the company’s quarterly and annual filings available on www.sedarplus.ca for
additional information on risks and uncertainties relating to the forward-looking statements. Sernova expressly
disclaims any intention or obligation to update or revise any forward-looking statements, whether as a result of
new information, future events or otherwise.
Press Release Communiqué de presse - Octobre 11, 2023 11 Octobre, 2023
Sernova to Provide Two Program Updates in Podium Presentations at 2023 IPITA Congress
Dr. Piotr Witkowski presenting new data from ongoing Phase 1/2 clinical trial of the Cell Pouch System™ in patients with type 1 diabetes (T1D)
Dr. Alice Tomei presenting preclinical data on transplantation of conformal coated islet cells in pre- vascularized Cell Pouch
LONDON, Ontario; WINDHAM COUNTY, Connecticut – October 10, 2023 – Sernova Corp. (TSX:SVA) (OTCQB:SEOVF) (FSE/XETRA:PSH), a clinical-stage company and leader in cell therapeutics, today announced two oral presentations at the upcoming International Pancreas and Islet Transplant Association (IPITA), International Xenotransplantation Association (IXA) Cell Transplant & Regenerative Medicine Association (CTRMA) joint congress taking October 26-29, 2023 in San Diego, CA.
More details about the conference, including accepted abstracts, are available on the IPITA website HERE. In alignment with the embargo policy, Sernova plans to share details from both speakers lectures following completion of their presentations.
Presenation details:
Abstract #: 105.1 Title: Islet allotransplantation into pre-vascularized Sernova Cell Pouch™ - Lessons learned from the first patient cohort Presenter: Piotr Witkowski, United States, Associate Professor of Surgery, University of Chicago Session: Islet Transplantation: Engineering the Islet Site Session Date and Time: Thursday, October 26, 2023 from 2:45 p.m. to 3:45 p.m. PT
Abstract #: 205.1 Title: Transplantation of conformal coated islets in a pre-vascularized Cell Pouch™ device for beta cell replacement in diabetic rats Presenter: Alice Tomei, United States, Associate Professor - Biomedical Engineering and Diabetes Research Institute, University of Miami Session: Engineering the site (Joint) Session Date and Time: Friday, October 27, 2023 from 8:30 a.m. to 9:30 a.m. PT
ABOUT SERNOVA CORP. AND THE CELL POUCH SYSTEM PLATFORM FOR CELL THERAPY Sernova Corp. is a clinical-stage biotechnology company that is developing therapeutic cell technologies for chronic diseases, including insulin-dependent diabetes, thyroid disease, and blood disorders that include hemophilia A. Sernova is currently focused on developing a ‘functional cure’ for insulindependent diabetes with its lead asset, the Cell Pouch System, a novel implantable and scalable medical device with immune protected therapeutic cells. On implantation, The Cell Pouch forms a natural vascularized tissue environment in the body for long-term survival and function of therapeutic cells that release essential factors that are absent or deficient in the bodies of patients with certain chronic diseases. Sernova’s Cell Pouch System has demonstrated its potential to be a ‘functional cure’ for people with T1D in an ongoing Phase 1/2 clinical study at the University of Chicago, with Cell Pouches implanted for periods of more than four years and counting.
Sernova is also advancing a proprietary technology in collaboration with the University of Miami to shield therapeutic cells from immune system attack with the goal to eliminate the need for chronic, systemic immunosuppression. In May 2022, Sernova and Evotec entered into a global strategic partnership to develop an implantable off-the-shelf iPSC (induced pluripotent stem cells) based islet replacement therapy. This partnership provides Sernova a potentially unlimited supply of insulinproducing cells to treat millions of patients with insulin-dependent diabetes (type 1 and type 2). Sernova continues to progress two additional development programs that utilize its Cell Pouch System: a cell therapy for hypothyroid disease resulting from thyroid gland removal and an ex vivo lentiviral Factor VIII gene therapy for hemophilia A.
FOR FURTHER INFORMATION, PLEASE CONTACT:
Christopher Barnes VP, Investor Relations Sernova Corp. christopher.barnes@sernova.com Tel: 519-902-7923 www.sernova.com
Investors: Corey Davis, Ph.D. LifeSci Advisors, LLC cdavis@lifesciadvisors.com Tel: 212-915-2577
Media: Hannah Holmquist LifeSci Communicaons hholmquist@lifescicomms.com Tel: 619-723-4326
FORWARD-LOOKING INFORMATION This release contains statements that, to the extent they are not recitations of historical facts, may constitute “forward-looking statements” that involve various risks, uncertainties, and assumptions, including, without limitation, statements regarding the prospects, plans, and objectives of the company. Wherever possible, but not always, words such as "expects", "plans", "anticipates", "believes", "intends", "estimates", "projects", "potential for" and similar expressions, or that events or conditions "will", "would", "may", "could" or "should" occur are used to identify forward-looking statements. These statements reflect management’s beliefs with respect to future events and are based on information currently available to management on the date such statements were made. Many factors could cause Sernova’s actual results, performances or achievements to not be as anticipated, estimated or intended or to differ materially from those expressed or implied by the forward-looking statements contained in this news release. Such factors could include, but are not limited to, the company’s ability to secure additional financing and licensing arrangements on reasonable terms, or at all; ability to conduct all required preclinical and clinical studies for the company’s Cell Pouch System and or related technologies, including the timing and results of those trials; ability to obtain all necessary regulatory approvals, or on a timely basis; ability to in-license additional complementary technologies; ability to execute its business strategy and successfully compete in the market; and the inherent risks associated with the development of biotechnology combination products generally. Many of the factors are beyond our control, including those caused by, related to, or impacted by the novel coronavirus pandemic. Investors should consult the company’s quarterly and annual filings available on www.sedarplus.ca for additional information on risks and uncertainties relating to the forward-looking statements. Sernova expressly disclaims any intention or obligation to update or revise any forward-looking statements, whether as a result of new information, future events or otherwise.
Subscribe to our Newsletter Abonnez-vous à notre newsletter
By subscribing to our Newsletter you will receive Sernova’s press releases and news as they develop by email. We do not send direct mail, customized online advertising, and we do not share your personal information. We respect your privacy. En vous abonnant à notre newsletter, vous recevrez par e-mail les communiqués de presse et les actualités de Sernova au fur et à mesure de leur développement. Nous n'envoyons pas de publipostage, de publicité en ligne personnalisée et nous ne partageons pas vos informations personnelles. Nous respectons votre vie privée.
Please read our Pour plus d’informations, veuillez lire notre Privacy policypolitique de confidentialité.







